Etymology The symbol for the planet Mercury (☿) has been used since ancient times to represent the element Hg are the most often studied NMR-active nuclei, having spins of 1⁄ 2 and 3⁄ 2 respectively. Hg occurs naturally in tiny traces as an intermediate decay product of 238 Most of the remaining radioisotopes have half-lives that are less than a day. Hg with a half-life of 444 years, and 203 There are seven stable isotopes of mercury, with 202 Mercury embrittlement is the most common type of liquid metal embrittlement, as mercury is a natural component of some hydrocarbon reservoirs and will come into contact with petroleum processing equipment under normal conditions. For this reason, mercury is not allowed aboard an aircraft under most circumstances because of the risk of it forming an amalgam with exposed aluminium parts in the aircraft. Since the amalgam destroys the aluminium oxide layer which protects metallic aluminium from oxidizing in-depth (as in iron rusting), even small amounts of mercury can seriously corrode aluminium. Mercury readily combines with aluminium to form a mercury-aluminium amalgam when the two pure metals come into contact. Sodium amalgam is a common reducing agent in organic synthesis, and is also used in high-pressure sodium lamps. Other elements that do not readily form amalgams with mercury include platinum. Several other first row transition metals with the exception of manganese, copper and zinc are also resistant in forming amalgams. Iron is an exception, and iron flasks have traditionally been used to transport the material. Mercury dissolves many metals such as gold and silver to form amalgams. Amalgams Mercury-discharge spectral calibration lamp Mercury reacts with solid sulfur flakes, which are used in mercury spill kits to absorb mercury (spill kits also use activated carbon and powdered zinc). Like silver, mercury reacts with atmospheric hydrogen sulfide. Mercury does not react with most acids, such as dilute sulfuric acid, although oxidizing acids such as concentrated sulfuric acid and nitric acid or aqua regia dissolve it to give sulfate, nitrate, and chloride. Table of thermal and physical properties of liquid mercury: Temperature (☌) Solid mercury is malleable and ductile and can be cut with a knife. Upon freezing, the volume of mercury decreases by 3.59% and its density changes from 13.69 g/cm 3 when liquid to 14.184 g/cm 3 when solid. This effect is due to lanthanide contraction and relativistic contraction reducing the radius of the outermost electrons, and thus weakening the metallic bonding in mercury. It has a freezing point of −38.83 ☌ and a boiling point of 356.73 ☌, both the lowest of any stable metal, although preliminary experiments on copernicium and flerovium have indicated that they have even lower boiling points. Compared to other metals, it is a poor conductor of heat, but a fair conductor of electricity. Mercury is a heavy, silvery-white metal that is liquid at room temperature. Properties Physical properties An old pound coin (density ~7.6 g/cm 3) floats on mercury due to the combination of the buoyant force and surface tension. Electricity passed through mercury vapor in a fluorescent lamp produces short-wave ultraviolet light, which then causes the phosphor in the tube to fluoresce, making visible light. It remains in use in scientific research applications and in amalgam for dental restoration in some locales. Mercury is used in thermometers, barometers, manometers, sphygmomanometers, float valves, mercury switches, mercury relays, fluorescent lamps and other devices, although concerns about the element's toxicity have led to the phasing out of such mercury-containing instruments. Exposure to mercury and mercury-containing organic compounds is toxic to the nervous system, immune system and kidneys of humans and other animals mercury poisoning can result from exposure to water-soluble forms of mercury (such as mercuric chloride or methylmercury) either directly or through mechanisms of biomagnification. The red pigment vermilion is obtained by grinding natural cinnabar or synthetic mercuric sulfide. Mercury occurs in deposits throughout the world mostly as cinnabar ( mercuric sulfide). A heavy, silvery d-block element, mercury is the only metallic element that is known to be liquid at standard temperature and pressure the only other element that is liquid under these conditions is the halogen bromine, though metals such as caesium, gallium, and rubidium melt just above room temperature. 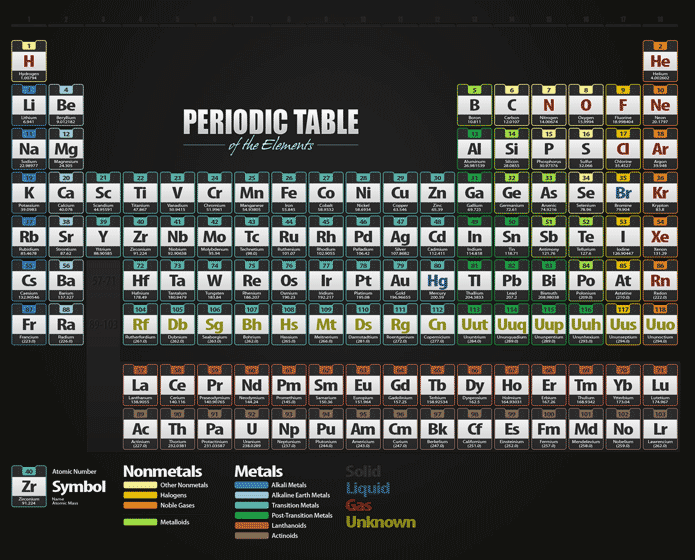
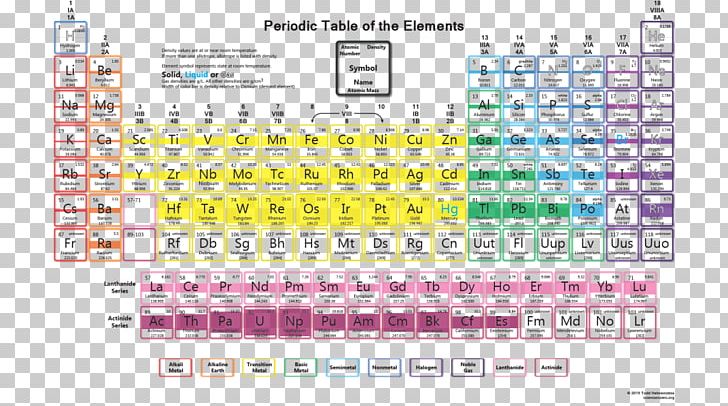
It is also known as quicksilver and was formerly named hydrargyrum ( / h aɪ ˈ d r ɑːr dʒ ər ə m/ hy- DRAR-jər-əm) from the Greek words hydor (water) and argyros (silver). Mercury is a chemical element it has symbol Hg and atomic number 80. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
